 The electronic configuration of Barium will be 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 5p6 6s2. Naturally occurring barium (56 Ba) is a mix of six stable isotopes and one very long-lived radioactive primordial isotope, barium-130, identified as being unstable by geochemical means (from analysis of the presence of its daughter xenon-130 in rocks) in 2001. How do you write the electron configuration for Barium? The electronic configuration of Barium will be 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 5p6 6s2. What is the electronic configuration of Barium 56? What is the boiling Point of Barium in Kelvin?īoiling Point of Barium in Kelvin is 2143 K. Rewrite and complete the following nuclear reaction by filling in the atomic number of Ba and mass number of Kr : 23592U+10n144.Ba+.36Kr+310n+Energy. Melting Point of Barium in Kelvin is 1000 K. What is the melting Point of Barium in Kelvin? What is the boiling Point of Barium?īoiling Point of Barium is 2143 K. Barium has 56 electrons out of which 2 valence electrons are present in the 6s2 outer orbitals of atom. How many valence electrons does a Barium atom have?īarium has 2 valence electrons. What is the color of Barium?īarium is of Silver color. The enhanced lines arising from the ionised atoms ( Ca +, Sr +, Ba + ) are. How to recognize an atomic number substituted text. 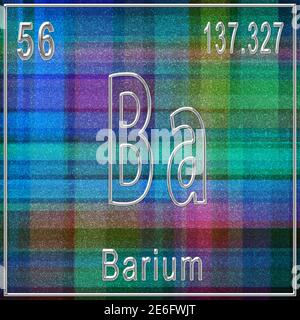
It is located in group 2 and period 6 in the modern periodic table. Tool to convert atomic numbers of the periodic classification of elements (or. Barium is the 56 element on the periodic table. 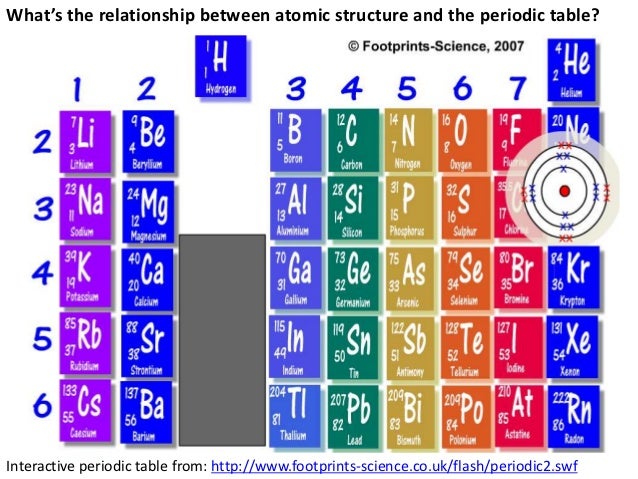
What is the position of Barium in the Periodic Table?īarium is a chemical element with the symbol Ba and atomic number 56. 
It is a soft, silvery-golden alkali metal with a melting point of 28.5 C (83.3 F), which makes it one of only five elemental metals that are liquid at or near room temperature. Barium is a chemical element with symbol Ba and atomic number 56. Caesium (IUPAC spelling cesium in American English) is a chemical element with the symbol Cs and atomic number 55. Barium occurs in 6 natural isotopes: 132 Ba, 134 Ba, 135 Ba, 136 Ba, 137 Ba and 138 Ba. Mass numbers of typical isotopes of Barium are 134-138. To form abbreviated notation of electronic configuration, the completely filled subshells are replaced by the noble gas of the preceding period in square brackets. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. The abbreviated electronic configuration of Barium is 6s2. Barium a soft, silvery very reactive metallic element is the fifth element in Group 2 of the periodic table, the alkaline earth elements.It has an atomic number of 56, atomic mass of 137.327, and chemical symbol of Ba. As shown in Diagram 1, the symbol for barium is Ba. What is the abbreviated electronic configuration of Barium? Other elements found in this group include magnesium ( atomic number-12), calcium (atomic number-20), and radium (atomic number- 88). BA274 (BAW274) British Airways Flight Tracking and History - FlightAware British Airways 274 BAW274 / BA274 Upgrade to see tail number. The electronic configuration of Barium is 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 5p6 6s2. What is the electronic configuration of Barium? Optical Properties of Barium Refractive IndexĪcoustic Properties of Barium Speed of Soundīarium Thermal Properties - Enthalpies and thermodynamics Refer to table below for the Electrical properties ofBarium Electrical Conductivityīarium Heat and Conduction Properties Thermal Conductivity Hardness of Barium - Tests to Measure of Hardness of Element Mohs Hardnessīarium is Conductor of electricity. Barium has many isotopes, but out of those isotopes, the isotope 138 Ba is the most abundant one (approximately 71.7 ). The crystal structure of barium is BCC (Body centered cubic). Check out our other guides on the game, like how many rules there are in total.Refer to below table for Barium Physical Properties Densityģ.51 g/cm3(when liquid at m.p density is $3.338 g/cm3) The atomic mass of barium is 137.33 u and its density is 3.51 g/cm 3, which is quite dense among all the other alkaline earth metals on the periodic table. That’s everything you need to know about how to beat RUle 18 in The Password game. However, this assumes the atom to exhibit a spherical shape, which is only obeyed for atoms in. The atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. It must be noted, atoms lack a well-defined outer boundary. You can change your Captcha if need be at any time. The atomic radius of Barium atom is 215pm (covalent radius). The symbol is an abbreviation for the element which may be obvious in some cases. Look out for the Captcha, which will probably have some elements that may conflict. Just make sure you don’t accidentally conflict with a previous rule now. 
Use any combination of these to ensure you complete Rule 18. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
